In a world where almost a third of the human population is overweight or obese, many of us strive daily to find a balance between health and indulgence. But what if there were a miracle pill which could allow us to indulge our sweet tooth guiltlessly? Well, sadly, there is no miracle pill but there is a miracle fruit, which can not only trick your taste buds but has also been wrapped in a conspiracy theory for almost half a century.
Synsepalum dulcificum – otherwise known as the miracle fruit or miracle berry – was first recorded in 1725 by French explorer Reynaud Des Marchais in West Africa. He noted that the locals would consume the fruit of this plant to make bland and sour breads taste sweet. However, the berry remained in relative obscurity until around the mid 20th century.

Miraculin
First identified in 1968 and sequenced in 1989 by Japanese scientists, miraculin is the taste modifying protein that gives the miracle fruit its defining property.
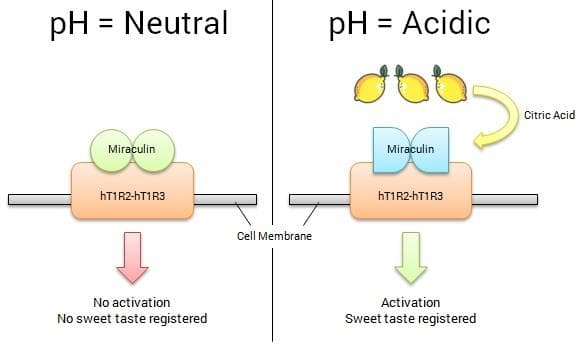
The human sense of taste is comprised of five basic tastes: sweet, salty, bitter, sour, and umami. Each of these is activated when agonist ligands bind to taste receptors on our taste bud cells. Here, agonist ligands are those which bind to, and cause a response by, our taste receptors. The human sweet taste receptor, hT1R2-hT1R3, is activated by such agonist ligands as the sweet-tasting proteins thaumatin (used widely in many foods and beverages) and brazzein (still awaiting FDA approval), and the sweet-tasting molecules aspartame, saccharin, and sucralose, which are used in artificial sweeteners. Unlike these proteins and molecules, at neutral pH miraculin acts as an antagonist to hT1R2-hT1R3, binding to the receptor and inhibiting binding of other sweet-tasting substances. So, while miraculin does not taste sweet itself, at neutral pH, it also dampens the sweetness of other substances. At
low pH levels (acidic), miraculin changes shape which allows for its active site to bind to the hT1R2 unit of the hT1R2-hT1R3 sweet receptor. This activates the receptor, and we register a sweet taste.
Miraculin is rather unusual, as it is structurally unrelated to the other ‘sweet proteins’ and unlike the more conventional sweet-tasting proteins, miraculin has a rather bland taste at neutral pH. Miraculin’s true taste-modifying properties are only exhibited when exposed to an acidic environment.
The Miralin Co. Conspiracy
In the 1960s, Robert Harvey, a biochemical postgraduate student, was introduced to miracle fruit. After contemplating the potential of the fruit, he decided to found the Miralin Company. Dedicated to providing a healthy alternative to both sugar and sweeteners, Miralin pushed forward with very successful product testing. However, Miralin’s luck was about to change, in what would become the Miralin Co. conspiracy.
In 1974, Harvey began to believe he was being followed from work to his home. After that, cars were seen driving past Miralin’s offices, with someone inside taking photographs. And then, one summer’s night, the Miralin Company’s office was burgled. In the middle of the ransacked office floor lay open Miralin’s FDA file.
Harvey expressed how good a relationship he and Miralin had had with the FDA and how his company had had their full support. However, on the eve of Miralin’s launch, the FDA suddenly banned the use of miraculin, labelling it as an additive. This meant that their product could not be sold as a sugar substitute without further, and very expensive, testing. Years of testing miraculin eventually bankrupted the Miralin Company and to this day the FDA has never overturned their ruling. Once again, the miracle fruit fell into obscurity; that is, until the turn of the 21st century.
Mass Production of Miraculin
In the past 20 years, interest has once again peaked around the miracle fruit. Miraculin may have the potential to compete with or even replace artificial sweeteners one day, which have been tentatively linked to other, more serious health issues and are often plagued with unpleasant aftertastes.
One thing which could stand in the way of miraculin becoming a competitive sweetener (other than regulatory approval) is simply producing enough of it. Miracle fruit is notoriously difficult to grow outside of its natural environment and its scarcity compared with high demand would likely make it too expensive for the average consumer. That’s where science steps in.
Often, the go-to method for mass production of a specific protein, such as recombinant insulin, is to artificially insert the gene for producing said protein into an organism, such as bacteria or yeast. That organism is grown in bioreactors and the protein of choice harvested in large quantities. This is exactly what was initially intended with organisms such as Escherichia coli and yeast. However, the recombinant miraculin harvested from these transgenic organisms lacked its sweetness-inducing activity. But why?
Professor Hiroshi Ezura and his lab at Tsukuba University have been investigating the production of miraculin for over a decade. In their research they proposed that specific post-translational modifications of miraculin may be necessary for its defining property of taste modification. Their hypothesis ultimately proved correct.
When a protein is being produced by an organism, the gene is ‘read’ and transcribed into mRNA. That mRNA is then translated by ribosomes and tRNA into a chain of amino acids. After this translation stage, modifications can be made to the amino acids, such as phosphorylation and glycosylation (and more). The important distinction here is that different organisms perform different post-translational modifications. A transgenic organism may not construct a protein exactly how it is in its native form when constructed by the original organism, and thus may not function exactly how it does in its native form.
The researchers at Tsukuba University found exactly this. Their research concluded that plant-specific N-glycosylation is necessary for miraculin with fully functional taste-modification properties. N-glycosylation is a post-translational modification where a glycan molecule is attached to an asparagine amino acid in a protein, which can affect folding and activity of the protein. As miraculin is produced in plants, Prof. Hiroshi Ezura and his team decided to use other plants as ‘factories’ to mass produce the miraculin outside of miracle fruit. They postulated that this should minimalise the differences in post-translational modifications. The team have created both transgenic lettuce and tomatoes, which express recombinant miraculin with functional taste-modification properties.
This is significant, as both lettuce and tomatoes are much more easily grown in all parts of the world and both yielded a high amount of miraculin. In fact, their research has steered solely towards transgenic tomatoes, as these produce near identical recombinant miraculin to the native, with near identical sweetness-inducing activity. What’s more, the miraculin gene has been shown to be stable: the progeny of the transgenic tomatoes are also able to produce miraculin at similar quantities to the parent plants (the lettuce progeny resulted in only around 10% production of miraculin when compared to the original parent line). This removes the need to repeatedly artificially insert the miraculin gene into future tomato seeds, which also reduces long-term cost of production. To top it all off, the transgenic tomatoes produce, on average, ten times more miraculin than that of natural miracle fruit.
Tsukuba University’s researchers still have a lot of testing in their future but, right now, their research is looking promising for viable, cost-effective mass production of miraculin.
The Future
Since 1975, just one year after the Miralin Company was set to launch, worldwide obesity has tripled and around 2 billion people are now overweight or obese. It’s difficult not to speculate what a world with almost half a century of miraculin-supplemented foods might have looked like today. However, we do not live in that world. So, what does this mean for our future?
Well, for now, not a whole lot. Until miraculin is generally recognised as safe and, further, recombinant miraculin from GM crops is recognised as safe (that is an entire thing unto itself), it is unlikely that miraculin can be a viable, cost-effective, and mass-producible replacement for sugar and current artificial sweeteners. Thus, miraculin will remain relegated to party tricks and niche cafés; at least, for now.



