Marchantia, or liverwort, mothers and fathers are haploid, carrying only one copy of their chromosome. When they fuse, they generate a diploid embryo with two copies of the genome, but things could go wrong when genomes conflict, causing the death of the offspring. A research team led by Prof. Berger at the Gregor Mendel Institute of Molecular Plant Biology (Vienna, AUSTRIA) have discovered that mothers and fathers don’t make an equal contribution to their offspring, at least in Marchantia. Their research, published in the scientific journal eLife, shows a sophisticated way of regulating genes that may have been happening for almost half a billion years.
The paper by Sean Akira Montgomery and colleagues describes a new mechanism of genomic imprinting, named “paternal chromosome repression”, that epigenetically controls the expression of genes inherited from the mother and the father in diploid embryos in Marchantia polymorpha.
As the Marchantia embryos are diploid, they have genes from both their mother and father. Yet, when the authors analyzed the transcriptomes (the set of RNA molecules expressed from an individual’s genes) thirteen days after fertilization, they found almost exclusive expression of genes from maternal origin.
By contrast, paternal genes are suppressed through a genomic imprinting mechanism that relies on the deposition of repressive marks (H3K27me3) that takes place shortly after fertilization. These molecular tags leave the father’s genes in the embryo but deactivate them, leaving them silent. The plant does this surprisingly rapidly to keep the embryo safe.
“We found that Marchantia completely inactivates the paternal chromosomes in the embryo, even before the fusion of the paternal and maternal genomes. This way, Marchantia maintains a functional haploidy even during the short stage in which it becomes diploidic,” said Sean Montgomery, the first author of the paper, in a press release.
The findings could help understand how land plants of today originated 470-450 million years ago from green algae that started terrestrial colonization by adapting to drought and ultraviolet radiation.
Scientists are using the liverwort Marchantia polymorpha to understand the evolution of land plants, as this plant diverged from other plants early after the emergence of the aquatic ancestor. This divergence makes it useful as a model organism to dissect the mechanisms that control the formation of the plant body, as well as the alternation of generations throughout the plant life cycle.
During the evolution of land plants, the life process transitioned from a haploid life cycle of green algae that have just one copy of their genome to a diploid life cycle of vascular plants that have two copies. Interestingly, the non-vascular plant Marchantia has a dominant haploid generation (a unicellular spore that develops into a multicellular body) and a diploid generation (an embryo formed upon fertilization of egg and sperm cells), as depicted in Figure 1. However, the mechanisms that control gene dosage (of maternal and paternal origin) during Marchantia’s life span are still elusive.
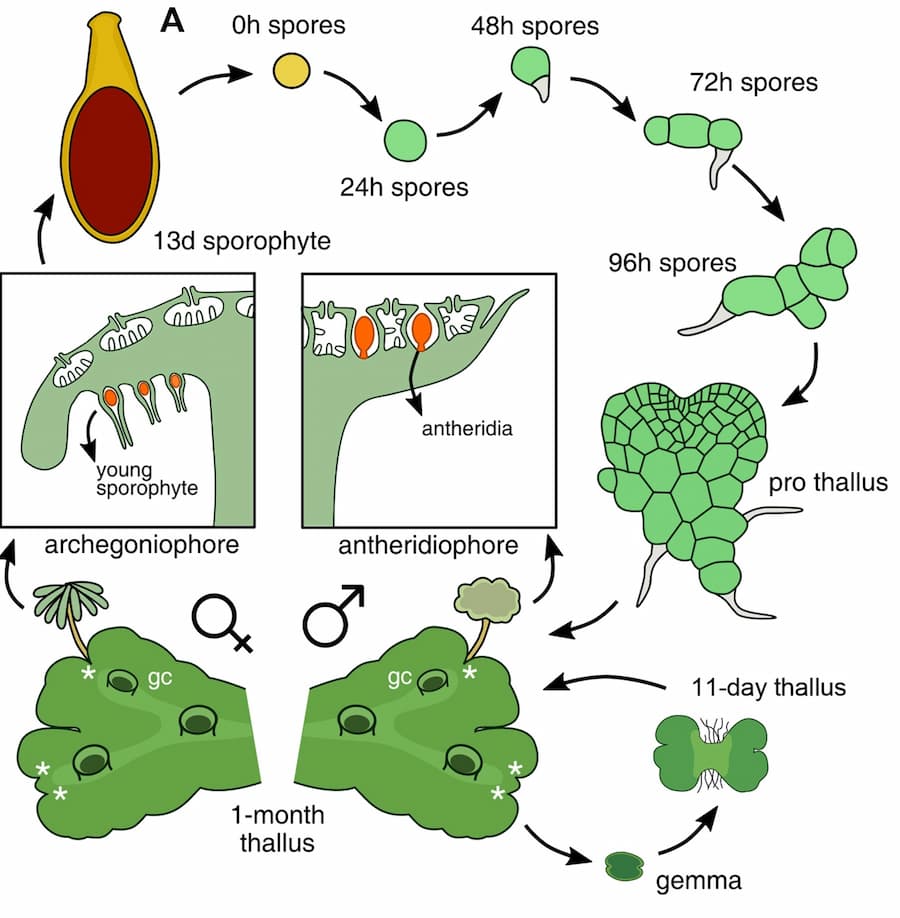
The research group found that silencing of the paternal genome is maintained during the diploid generation, but repressive chromatin modifications are reversed in the following haploid generation.
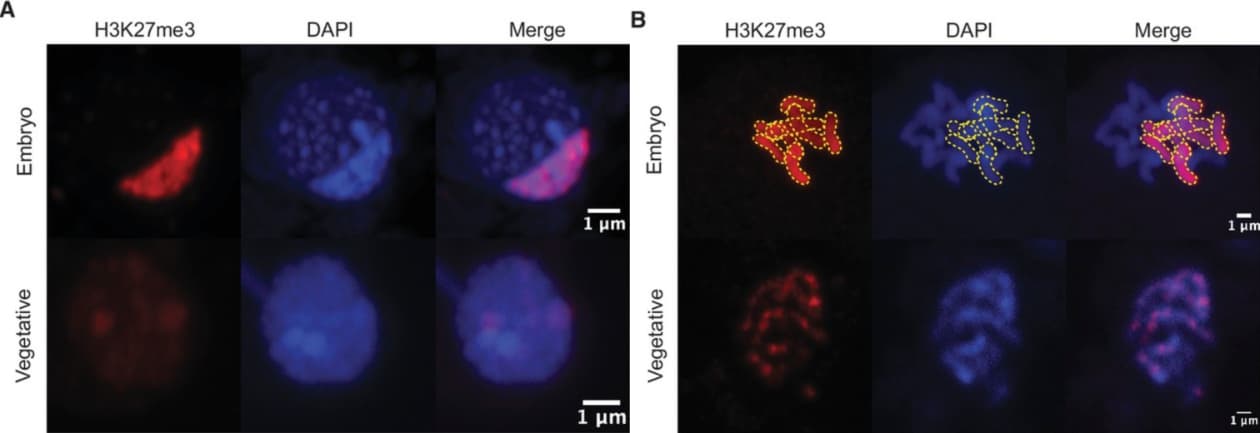
To visualize repressive marks in nuclei of Marchantia cells at different developmental stages, the authors employed immunofluorescence techniques (Fig. 2). They found that diploid embryos showed large heterochromatic regions during the interphase and dense coating of chromosomes during the mitotic phase. By contrast, haploid vegetative cells only showed irregular patterns of H3K27me3.
To understand the biological significance of paternal chromosome repression, the team generated mutants that were impaired in this mechanism. Although expression of alleles of paternal origin was re-established, mutant embryos displayed severe growth defects and a very high mortality rate at maturity. In addition, those few embryos that survived could not produce viable spores, strongly suggesting that silencing the paternal genome in the diploid generation is critical to ensure the propagation of the species (through asexual and sexual reproduction).
Intriguingly, the proper development of embryos is determined by genes inherited from both the mother and the father in several eukaryotic organisms but only by maternal genes in Marchantia.
“The embryo development depends solely on the expression of the maternal genes. In a way, the maternal genes have total control. Disrupting this process leads to the expression of the paternal genes and the death of the embryo,” explains Frédéric Berger, senior author of the paper and leader of the research group investigating chromatin architecture and function in plants.
Genomic imprinting has already been described in mammals and flowering plants with a dominant diploid generation. Still, it is the first time that this regulatory mechanism has been reported in a eukaryotic organism such as M. polymorpha that alternates a dominant haploid generation with a short diploid embryonic generation.
This study opens up the possibility that different eukaryotic organisms might adopt different strategies to regulate gene dosage to guarantee genome stability and the successful reproduction of the species.
ORIGINAL ARTICLE
Montgomery, S.A., Hisanaga, T., Wang, N., Axelsson, E., Akimcheva, S., Sramek, M., Liu, C. and Berger, F. (2022) “Polycomb-mediated repression of paternal chromosomes maintains haploid dosage in diploid embryos of Marchantia,” eLife, 11. https://doi.org/10.7554/eLife.79258






