
The Bromeliaceae family includes Spanish moss, pineapples, and air plants. Up to 2/3 of Bromeliaceae species are threatened with extinction due to habitat loss and fragmentation, climate change, invasive species, and commercialization for ornamental purposes. Understanding the reproductive effort, which is the trade-off between devoting resources to reproduction versus individual growth, of Bromeliaceae can provide insight into conservation strategies. A new study published in in silico Plants provides a novel mathematical model of Bromeliaceae growth and reproductive effort.
Erin Bodine, Associate Professor of Mathematics at Rhodes College, and colleagues modified a dynamic plant growth model previously developed to account for the timing and allocation of resources in both sexual reproduction (via flowers) and asexual reproduction (via genetically identical clonal offshoots). Using the results of model simulations, they then calculated the reproductive effort over the lifetime of a bromeliad using existing and novel measures for effort that account for allocation towards asexual reproduction in different ways.
Calculating reproductive effort for Bromeliaceae is challenging because of the existence of two modes of reproduction: sexual and asexual. The leaves of bromeliads grow in a spiral rosette pattern. New leaves are generated at the center from apical meristems. Vegetative growth of leaves and stem continues until a critical size, developmental, or environmental cues trigger a transition to reproductive growth (see vegetative growth in figure 1). At this point, the apical meristem will generally convert to produce a single cluster of flowers (an inflorescence). When this structure is complete, the apical meristem will cease all activity and no further development and growth happens along that axis (see inflorescence growth in figure 1). Above the base of every leaf in the rosette there is also an axillary meristem which, if initiated, will produce a genetically identical clonal offshoot called a pup (see pup growth in figure 1). Each pup has an apical meristem capable of producing an inflorescence. Bromeliads capable of producing pups have an iteroparous reproductive strategy where individuals reproduce sexually multiple times from the inflorescences of multiple iterative pups. Bromeliads lacking the capability to produce pups have a semelparous reproductive strategy in which they undergo a one-time and subsequently lethal sexual reproductive event.
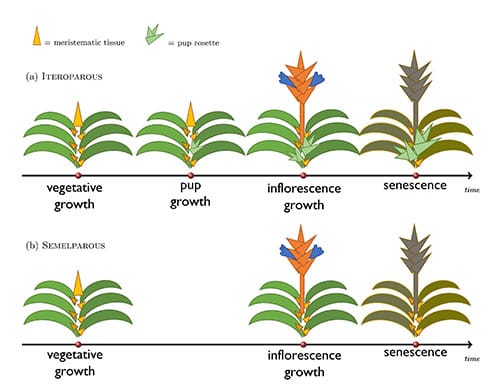
While plants of both strategies produce inflorescences, iteroparous plants are capable of producing multiple inflorescences. To allow for a direct comparison, the authors model the development of a single rosette until senescence assuming comparable rates of developmental timing and productivity in both an iteroparous and semelparous species. The mass of the pups of the iteroparous species are accounted for, but only until the senescence of the mother rosette. Across the Bromeliaceae family there exists a wide continuum of life cycle duration so the authors simulated a short time horizon of two years, and a long time horizon of nine years.
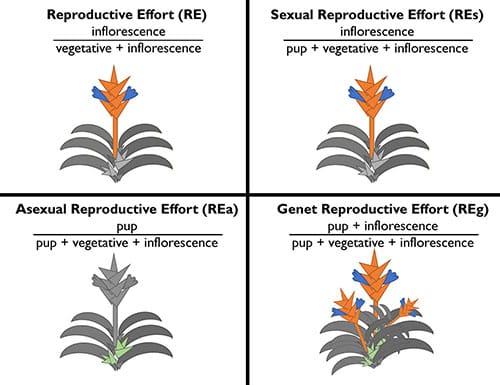
The authors consider four different formulations of reproductive effort (RE), each temporally dependent, changing as the rosette grows. The authors quantify the classic construction of reproductive effort of semelparous and iteroparous taxa as
- Reproductive Effort (RE) ∝ inflorescence mass divided by the sum of vegetative and reproductive mass of rosette
In this measure, the mass of pups for the iteroparous taxa are not included.
The authors propose three additional formulations of reproductive effort for iteroparous that consider pup mass as either vegetative or reproductive:
- Sexual Reproductive Effort (REs) ∝ inflorescence mass divided by the sum of pup, vegetative, and inflorescence mass of rosette
- Asexual Reproductive Effort (REa) ∝ pup mass divided by the sum of pup, vegetative, and inflorescence mass of rosette
- Genet Reproductive Effort (REg) = Sexual Reproductive Effort (REs) + Asexual Reproductive Effort (REa)
The final formulation represents an extension the calculation of reproductive effort for iteroparous taxa over the lifetime of a single genetic individual with multiple pups and inflorescences (a genet) using the classic construction of reproductive effort calculated at the senescence of each genetically identical rosette.
The simulations demonstrate that how pups are accounted for in the formulation of reproductive effort for iteroparous taxa will determine whether the semelparous or iteroparous taxa exhibits higher reproductive effort.
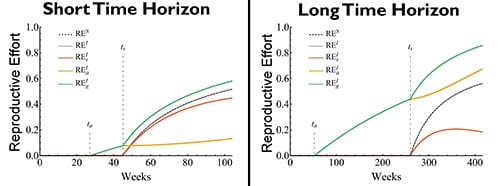
For short horizon taxa, when reproductive effort is calculated over the lifespan of a rosette, semelparous reproductive effort is greater than iteroparous when pup mass is accounted for as if it were purely vegetative in iteroparous taxa (compare REs vs REa and REs in figure 3). However, if pup and inflorescence mass are included in reproductive mass, as for the genet, then iteroparous reproductive effort is greater (compare REs and REg vs REs in figure 3).
Similarly, for long horizon taxa, because pups add and sustain mass for the genet for several years, iteroparous reproductive efforts are higher than semelparous if pup mass is included in reproductive mass, as for the genet and asexual reproduction (compare REg and REa vs REs in figure 3).
The authors conclude, “if differences in life history (i.e., iteroparous or semelparous) correspond to differences in reproductive effort, then understanding and quantifying these differences may provide valuable insights when constructing strategies aimed to conserve these and other imperiled bromeliad species.”
READ THE ARTICLE:
Bodine, E. N., Bush, C., Capaldi, A., & Jabaily, R. S. (2023). Modelling differences in reproductive effort between iteroparous and semelparous reproductive strategies in Bromeliaceae. in Silico Plants, 5(1). https://doi.org/10.1093/insilicoplants/diac019



