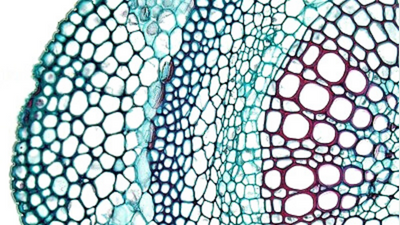
Primary growth arises from cell division at the tips of stems and roots causing them to elongate. The region of primary cell division is called the apical meristem. Three types of primary meristems arise from the apical meristem: the protoderm, ground meristem, and procambium. The procambium gives rise to vascular tissue (primary xylem and phloem), and the cambium.
Secondary growth is characterized by an increase in thickness of the plant. It is caused by cell division in the cambium. As cells in the cambium divide, they differentiate into secondary phloem towards the outside or secondary xylem towards the inside of the stem or root.
Studies point to one important transcription factor, MONOPTEROS (MP), in the role of cambium development. Curiously, there are two contradictory roles for MP:
- Promoter: During the transition from primary to secondary growth, MP activates PXY expression. Then PXY interacts with TDIF, forming a complex (TDIF-PXY) that promotes initial cambial cell divisions.
- Repressor: When secondary growth is established, MP represses cambial cell division.
PhD Student Kristine Bagdassarian (Durham University), under the supervision of Dr Natasha Savage (University of Liverpool), hypothesized that the seemingly contradictory role of MP might be integrated into the same network as a negative feedback loop. Together with other colleagues at Durham, the team present their work in the journal in silico Plants.
“Although the evidence for MP function were made at different plant life stages, it seemed unlikely to us that the underlying network would be that different, as the tissue organization at these stages is so similar. Consequently, we tested what would happen if both functions assigned to MP were present in a negative feedback loop” explains Savage.
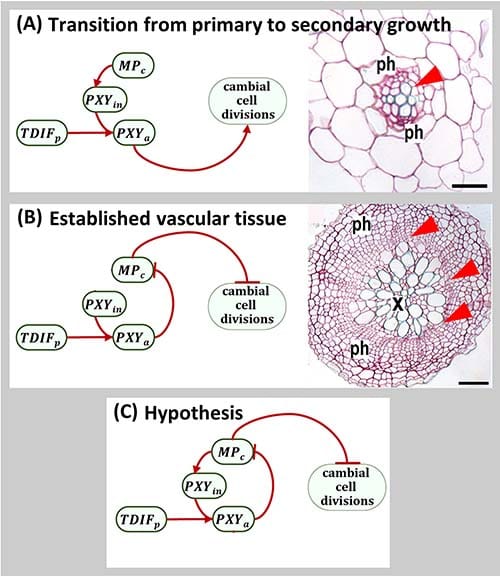
The authors developed a mathematical model to verify their MP negative feedback hypothesis. The model contained components which impact vascular tissue patterning: key regulatory hormones for plant growth and development (cytokinin and auxin), the proteins responsible for auxin transport, MP, PXY, and TDIF.
The model was run with and without the proposed negative feedback loop in place. The simulated auxin and cytokinin concentrations were compared to the observed concentrations in the xylem, cambium, and phloem. They expected to find cytokinin to be highest in the phloem and lowest in the xylem, and auxin to be highest in the cambium.
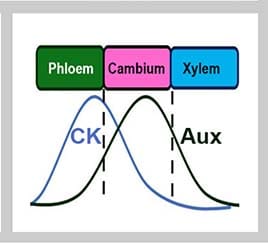
They discovered that including the negative feedback loop in the model increased the ability of the model to reproduce the highest relative amount of auxin in the cambium. Relative cytokinin concentrations in the three tissues were not affected by including or excluding the negative feedback loop.
The authors then investigated the role of the MP negative feedback loop in relation to the innate stability of MP. To do this, they altered MP degradation rate, which is a process by which the MP protein is broken down resulting in a reduced concentration of MP. They found that as the degradation of MP is reduced, and MP stability increased, the negative feedback loop became increasingly important for the modelled tissue to achieve the highest relative amount of auxin in the cambium.
These results support the notion that the MP negative feedback loop increases the ability of the system to pattern correctly.
The authors conclude, “cell division in the cambium drives wood formation. Wood is a versatile biomaterial and carbon sink. By understanding these interactions, we may be able to develop new strategies for manipulating wood formation to increase forest productivity and carbon capture.”
READ THE ARTICLE:
Kristine S Bagdassarian, J Peter Etchells, Natasha S Savage, A mathematical model integrates diverging PXY and MP interactions in cambium development, in silico Plants, 2023; diad003, https://doi.org/10.1093/insilicoplants/diad003
Data Availability
All numerical solutions presented in the results section, and all codes used to solve and analyze the numerical solutions, are freely available on GitHub at https://github.com/KristineBagdassarian/





